CLIA-Certified and
CAP-Accredited Laboratory
Our Vision: Delivering the Future of Healthcare
Pangea Laboratory is strongly committed to maintaining the highest standards of clinical laboratory testing. Since 2017, we have been certified by the Clinical Laboratory Improvement Amendments (CLIA) and accredited by the College of American Pathologists (CAP) to perform high-complexity testing. We are proud to have continually maintained our accreditation since then, using the latest testing technologies and keeping up with advances in best practices in an ever-improving industry.
Why CLIA Certification and CAP Accreditation Matters
Quality standards matter, especially when it comes to providing accurate, reliable, and timely information for healthcare diagnostics, treatment, or prevention. We adhere to the highest national and international standards so you can have confidence in our laboratory’s practices and results.
CLIA
In 1988, Congress passed CLIA to establish these standards for all non-research laboratory testing on human specimens. Any laboratory performing these tests must be certified by the Secretary of the Department of Health and Human Services (DHHS), with the Centers for Medicare & Medicaid Services (CMS) administering this program together with the Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA).

CLIA quality standards are specified for not just how the laboratory is administered, but also personnel qualifications and the analytic systems used. CLIA standards are national and apply to all laboratories that offer clinical laboratory services, and regulations depend on the complexity of the testing methods. Pangea Laboratory is certified for performing high complexity tests, and the requirements for this are the most stringent required.
We believe in going above and beyond, which is why beyond our CLIA certification, we are also a CAP accredited facility, appropriately licensed to safely perform testing on human and animal specimens. CAP inspections are conducted by expert teams of trained and practicing laboratory professionals across multiple disciplines every two years, which ensures that we meet or exceed industry-specific standards for laboratory operations.
CAP
CAP accreditation is internationally recognized as a means of demonstrating high-quality laboratory practices, and of meeting the most comprehensive scientifically endorsed standards. CAP requirements meet or exceed CLIA compliance requirements, and the exacting evaluations ensure high accuracy of testing results.

CAP-accredited laboratories must fulfill the requirements set out in their extensive checklists, which assess laboratories against thousands of detailed requirements every year. These requirements change annually to incorporate the most up-to-date best practices. It is a rigorous and stringent process that helps to ensure high standards of testing and care in areas like:
- Validation of test systems
- Analytic quality control
- Quality management of pre- and post-analytic processes
- Proficiency testing
- Communication to various stakeholders – clinicians, patients, administration
The CAP Accreditation Cycle
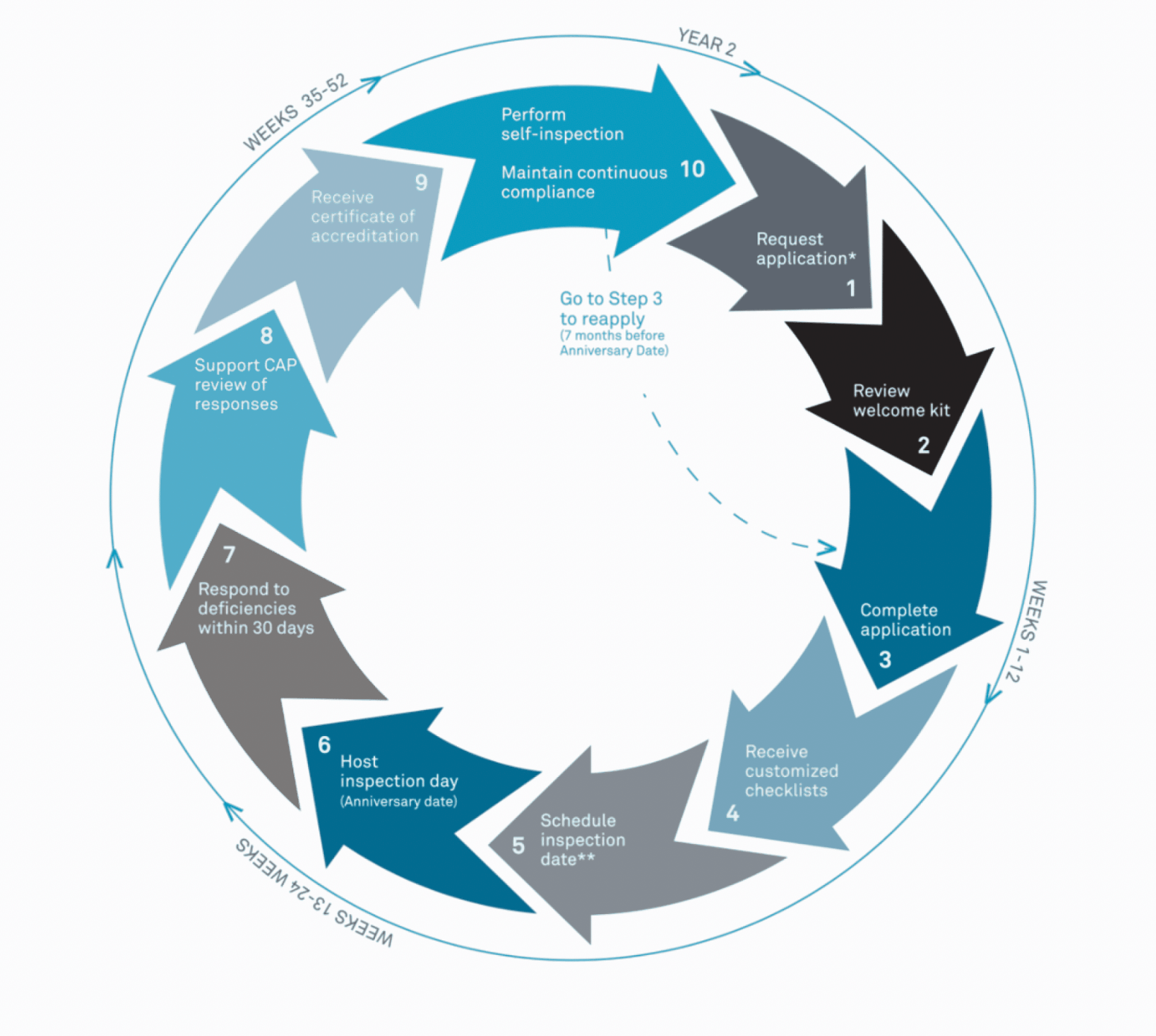
CAP’s Laboratory Accreditation Program is a dynamic process that fosters consistent improvement. All US inspections are unannounced after the initial inspection. To remain accredited, labs need to maintain continuous compliance with CAP’s requirements. We are proud to have a team that consistently and continually works towards improving our quality standards to stay in line with CAP best practices.
Learn About Our Products
Trust Pangea Laboratory for All Your Testing Needs
As a CLIA-certified and CAP-accredited lab, Pangea Laboratory meets and exceeds industry standards for clinical laboratory testing. We provide the highest service standards and offer the quality you can trust in our testing processes and results.
Choose Pangea Laboratory for all your molecular testing and Next Generation Sequencing needs. Contact us today at (949) 346-9393 to learn more about our custom testing solutions.
